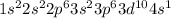Chemistry, 21.09.2019 14:30 TashaKessler3355
Give the actual ground-state electron configuration for copper (cu) using the complete form.
express the electron configuration using superscripts where appropriate. for example, the configuration for li would be entered as 1s^22s^1.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 22:00
Pls ill give u brainliest which of the following is true about science? 1. political conditions are unable to influence it. 2. economic concerns may prevent it from solving problems.
Answers: 2


Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Give the actual ground-state electron configuration for copper (cu) using the complete form.
e...
e...
Questions





English, 07.11.2019 02:31

Mathematics, 07.11.2019 02:31


Biology, 07.11.2019 02:31


History, 07.11.2019 02:31

Mathematics, 07.11.2019 02:31



Mathematics, 07.11.2019 02:31



Physics, 07.11.2019 02:31

Mathematics, 07.11.2019 02:31

English, 07.11.2019 02:31