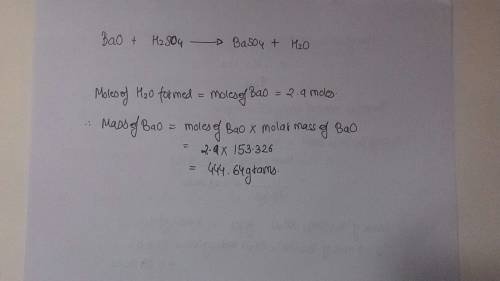Chemistry, 24.01.2020 17:31 southchrista3
Using the formula bao + h2so4 = baso4 + h2o if 2.9 miles of water is formed during this reaction then how many grams of bao was used?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1
You know the right answer?
Using the formula bao + h2so4 = baso4 + h2o if 2.9 miles of water is formed during this reaction the...
Questions

Social Studies, 18.12.2019 05:31




Physics, 18.12.2019 05:31

Mathematics, 18.12.2019 05:31

Biology, 18.12.2019 05:31


Biology, 18.12.2019 05:31

Mathematics, 18.12.2019 05:31


Social Studies, 18.12.2019 05:31

Mathematics, 18.12.2019 05:31


History, 18.12.2019 05:31


Mathematics, 18.12.2019 05:31



Mathematics, 18.12.2019 05:31