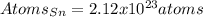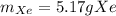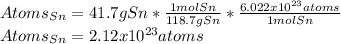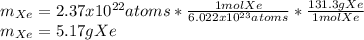Chemistry, 18.09.2019 09:10 justyne2004
How many atoms are there in 41.7g of tin.(sn)? what is the mass of 2.37•10^22 atoms of xenon(xe) gas?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
How many atoms are there in 41.7g of tin.(sn)? what is the mass of 2.37•10^22 atoms of xenon(xe) ga...
Questions


Advanced Placement (AP), 29.01.2022 22:00

Social Studies, 29.01.2022 22:00


Mathematics, 29.01.2022 22:10

Health, 29.01.2022 22:10


Computers and Technology, 29.01.2022 22:10




Medicine, 29.01.2022 22:10



Social Studies, 29.01.2022 22:10

Mathematics, 29.01.2022 22:10


Mathematics, 29.01.2022 22:10


Mathematics, 29.01.2022 22:10