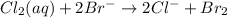Which best describes the oxidizing agent in this reaction?
cl2(aq) + 2brmc003-1.jpg(aq) mc00...

Chemistry, 07.10.2019 00:00 phillipsk5480
Which best describes the oxidizing agent in this reaction?
cl2(aq) + 2brmc003-1.jpg(aq) mc003-2.jpg 2clmc003-3.jpg(aq) + br2(aq)
bromine (br) is the oxidizing agent because it gains an electron.
bromine (br) is the oxidizing agent because it loses an electron.
chlorine (cl) is the oxidizing agent because it gains an electron.
chlorine (cl) is the oxidizing agent because it loses an electron.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

Chemistry, 23.06.2019 10:00
The temperature of a lead fishing weight rises from 26 °c to 38 °c as it absorbs 11.3 j of heat. what is the mass of the fishing weight in grams?
Answers: 1
You know the right answer?
Questions

History, 10.02.2021 14:00




Mathematics, 10.02.2021 14:00

History, 10.02.2021 14:00


Biology, 10.02.2021 14:00




Mathematics, 10.02.2021 14:00

English, 10.02.2021 14:00

History, 10.02.2021 14:00


English, 10.02.2021 14:00


Mathematics, 10.02.2021 14:00

Mathematics, 10.02.2021 14:00

SAT, 10.02.2021 14:00