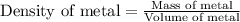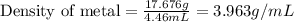Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
Apiece of metal (mass = 17.676 g) is placed in 11.00 ml of chloroform (d = 1.498 g/ml) in a 25-ml gr...
Questions

Biology, 01.10.2019 18:20


Chemistry, 01.10.2019 18:20





Chemistry, 01.10.2019 18:20




Computers and Technology, 01.10.2019 18:20








Mathematics, 01.10.2019 18:20