Identify the class of organic compounds to which the product of this reaction belongs
...

Chemistry, 02.11.2019 23:31 kellyzeissss4181
Identify the class of organic compounds to which the product of this reaction belongs
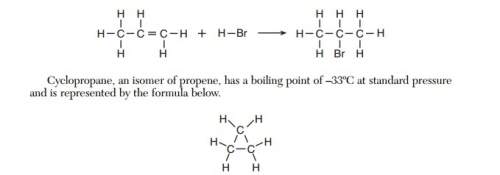

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

You know the right answer?
Questions

English, 05.12.2019 11:31

Biology, 05.12.2019 11:31


Arts, 05.12.2019 12:31

History, 05.12.2019 12:31

History, 05.12.2019 12:31

Advanced Placement (AP), 05.12.2019 12:31

Business, 05.12.2019 12:31




Mathematics, 05.12.2019 12:31

Mathematics, 05.12.2019 12:31

Mathematics, 05.12.2019 12:31




English, 05.12.2019 12:31

Mathematics, 05.12.2019 12:31

Mathematics, 05.12.2019 12:31



