
Chemistry, 29.09.2019 13:10 Kingmoney959
According to hess’s law, if a series of intermediate reactions are combined, the enthalpy change of the overall reaction is
the difference between the enthalpies of the intermediate reactions.
the sum of the enthalpy changes of the intermediate reactions.
the product of the enthalpy changes of the intermediate reactions.
the fraction of the individual enthalpies of the intermediate reactions.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1
You know the right answer?
According to hess’s law, if a series of intermediate reactions are combined, the enthalpy change of...
Questions





Social Studies, 08.12.2021 17:40

Mathematics, 08.12.2021 17:40

Mathematics, 08.12.2021 17:40


History, 08.12.2021 17:40



Physics, 08.12.2021 17:40



Social Studies, 08.12.2021 17:40

Mathematics, 08.12.2021 17:40


Biology, 08.12.2021 17:40


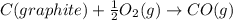
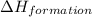
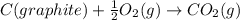
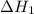
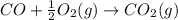
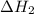
![\Delta H_{formation}=[n\times \Delta H_1]+[n\times \Delta H_2]](/tpl/images/0273/8162/6e820.png)


