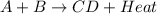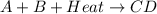Chemistry, 04.02.2020 06:06 adstone814
When the products of a reaction are hotter than the reactants: the reaction is exothermic. the reaction is endothermic. the reactants lost internal energy. the change in enthalpy is positive.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1


Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1

Chemistry, 22.06.2019 22:00
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1
You know the right answer?
When the products of a reaction are hotter than the reactants: the reaction is exothermic. the reac...
Questions




Mathematics, 18.08.2021 05:10


Mathematics, 18.08.2021 05:10


Mathematics, 18.08.2021 05:10



Mathematics, 18.08.2021 05:10

Biology, 18.08.2021 05:10


Biology, 18.08.2021 05:10



Chemistry, 18.08.2021 05:10

Mathematics, 18.08.2021 05:10

Mathematics, 18.08.2021 05:10

Mathematics, 18.08.2021 05:10