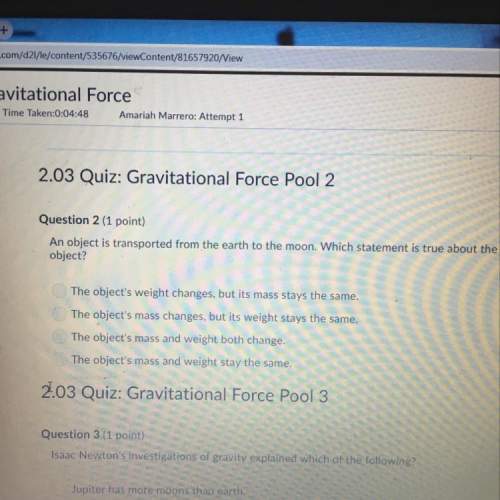
Which property determines an atom's ability to attract electrons shared in a chemical bond?
a...

Chemistry, 06.10.2019 01:00 almasahagung
Which property determines an atom's ability to attract electrons shared in a chemical bond?
a. ionization energy
b. atomic radius
c. electronegativity
d. ionic radius

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
Questions

History, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Biology, 10.09.2020 23:01

Chemistry, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Geography, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01

Mathematics, 10.09.2020 23:01