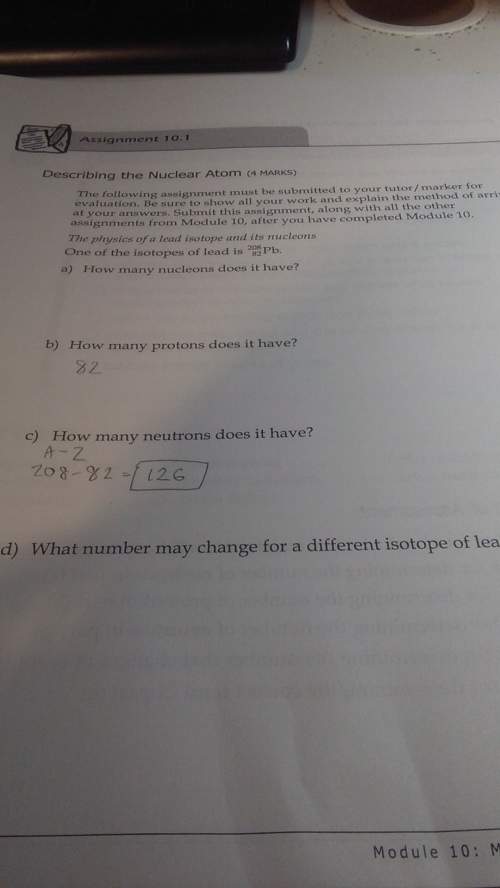With a and d? i've done b and c already
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
Questions

Mathematics, 24.06.2021 01:00

English, 24.06.2021 01:00

Mathematics, 24.06.2021 01:00

Mathematics, 24.06.2021 01:00

Mathematics, 24.06.2021 01:00



Mathematics, 24.06.2021 01:00


Mathematics, 24.06.2021 01:00


Mathematics, 24.06.2021 01:00



Mathematics, 24.06.2021 01:00

Mathematics, 24.06.2021 01:00

Mathematics, 24.06.2021 01:00



Mathematics, 24.06.2021 01:00