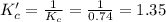Chemistry, 19.04.2021 18:20 jeremiaht7
At some temperature for the equilibrium PX3(g) + X2(g) # PX5(g) the equilibrium constant is 0.74. At the same temperature the equilibrium constant for PX5(9) + PX3(g) X2(g) is

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1


Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3
You know the right answer?
At some temperature for the equilibrium PX3(g) + X2(g) # PX5(g) the equilibrium constant is 0.74. At...
Questions

English, 17.12.2020 23:40

Mathematics, 17.12.2020 23:40

Chemistry, 17.12.2020 23:40



Mathematics, 17.12.2020 23:40

Mathematics, 17.12.2020 23:40




Chemistry, 17.12.2020 23:40





Spanish, 17.12.2020 23:40

Mathematics, 17.12.2020 23:40



Mathematics, 17.12.2020 23:40

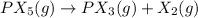 is 1.35
is 1.35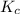
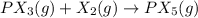
![K_c=\frac{[PX_5]^1}{[PX_3]^1[X_2]^1}](/tpl/images/1268/5086/e23fb.png)
![0.74=\frac{[PX_5]^1}{[PX_3]^1[X_2]^1}](/tpl/images/1268/5086/ef131.png)
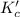 is written as:
is written as:
![K_c'=\frac{[PX_3]^1[X_2]^1}{[PX_5]^1}](/tpl/images/1268/5086/99777.png)