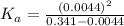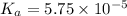Chemistry, 19.04.2021 16:10 NarutoBeast8049
In the laboratory, a general chemistry student measured the pH of a 0.341 M aqueous solution of benzoic acid, C6H5COOH to be 2.351. Use the information she obtained to determine the Ka for this acid.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

You know the right answer?
In the laboratory, a general chemistry student measured the pH of a 0.341 M aqueous solution of benz...
Questions



Mathematics, 03.08.2020 14:01


Social Studies, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01



Mathematics, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01


Mathematics, 03.08.2020 14:01





English, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01


Mathematics, 03.08.2020 14:01

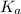 for the acid is
for the acid is 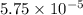
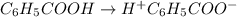
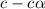
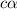
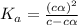
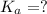
![pH=-log[H^+]](/tpl/images/1268/1073/15713.png)
![[H^+]=10^{-2.351}=0.0044](/tpl/images/1268/1073/85ea6.png)
![[H^]=c\alpha=0.0044](/tpl/images/1268/1073/4f06a.png)