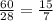Silicon can be made by heating a mixture of sand (silicon dioxide) with magnesium
powder.
The...

Silicon can be made by heating a mixture of sand (silicon dioxide) with magnesium
powder.
The equation for this reaction is shown below.
SiO2 (s)+ 2Mg (s) → 2MgO (s) + Si (s)
Calculate the mass of silicon dioxide needed to make 1 g of silicon.
Relative atomic masses: O = 16; Si = 28
Mass = ?

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 23.06.2019 06:30
1.17 mol hcl and 2.5 mol naoh react according to the equation hcl + naoh -> nacl + h2o . if the limiting reactant is hcl, determine the amount of excess reactant that remains. answer in units of mol.
Answers: 1
You know the right answer?
Questions


Advanced Placement (AP), 22.07.2019 23:30








History, 22.07.2019 23:30



Chemistry, 22.07.2019 23:30

Mathematics, 22.07.2019 23:30



Mathematics, 22.07.2019 23:30

History, 22.07.2019 23:30