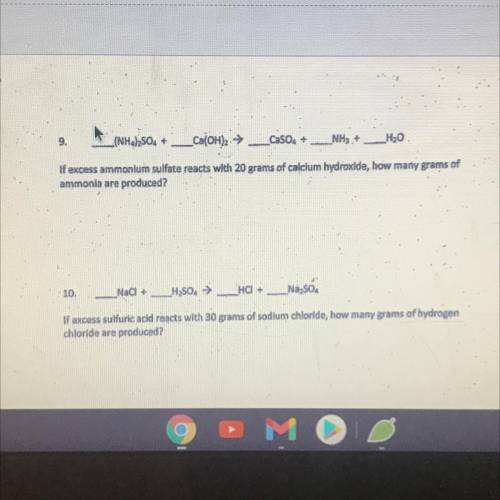
Help please due on 4/17
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

Chemistry, 23.06.2019 12:30
17) large amounts of very important metal titanium are made by reacting magnesium metal with titanium tetrachloride. titanium metal and magnesium chloride are produced. a) write the balanced equation for this reaction. b) how many kilograms of magnesium are required to produce 1.00 kilograms of titanium? ( show work, .)
Answers: 1

Chemistry, 23.06.2019 12:30
)a children’s liquid cold medicine has a density of 1.23 g/ml. if a child is to take 2.5 tsp in a dose, what is the mass in grams of this dose? (1 tsp = 5 ml)
Answers: 1
You know the right answer?
Questions


Mathematics, 19.07.2019 13:00

Biology, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00

Mathematics, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00

Biology, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00

Mathematics, 19.07.2019 13:00

Biology, 19.07.2019 13:00

Biology, 19.07.2019 13:00

Biology, 19.07.2019 13:00




Social Studies, 19.07.2019 13:00

Social Studies, 19.07.2019 13:00