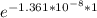Chemistry, 16.04.2021 01:00 BreBreDoeCCx
g Consider (12.5 A) micro-grams of a radioactive isotope with a mass number of (78 B) and a half-life of (32.6 C) million years. If energy released in each decay is 32.6 keV, determine the total energy released in joules (J) in 1 (one) year. Give your answer with three significant figures.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
g Consider (12.5 A) micro-grams of a radioactive isotope with a mass number of (78 B) and a half-lif...
Questions

Mathematics, 05.07.2019 12:30



Mathematics, 05.07.2019 12:30

Mathematics, 05.07.2019 12:30







English, 05.07.2019 12:30

Mathematics, 05.07.2019 12:30




Social Studies, 05.07.2019 12:30

History, 05.07.2019 12:30

Mathematics, 05.07.2019 12:30

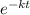
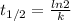
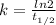
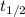 = 32.6 + 18 = 50.6 × 10⁶ years
= 32.6 + 18 = 50.6 × 10⁶ years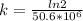 = 1.361 × 10⁻⁸ year⁻¹
= 1.361 × 10⁻⁸ year⁻¹