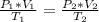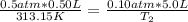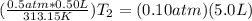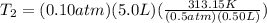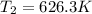Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1

Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3
You know the right answer?
A gas has a volume of 0.50 L, a pressure of 0.5 atm, and a temperature of 40°C. What will be the new...
Questions


English, 24.06.2020 01:01

Health, 24.06.2020 01:01

English, 24.06.2020 01:01

Mathematics, 24.06.2020 01:01

Social Studies, 24.06.2020 01:01


Mathematics, 24.06.2020 01:01

Mathematics, 24.06.2020 01:01




Mathematics, 24.06.2020 01:01


Social Studies, 24.06.2020 01:01

Health, 24.06.2020 01:01

History, 24.06.2020 01:01

Computers and Technology, 24.06.2020 01:01

Mathematics, 24.06.2020 01:01