
Chemistry, 15.04.2021 01:20 raffaldarmaki9412
HURRY
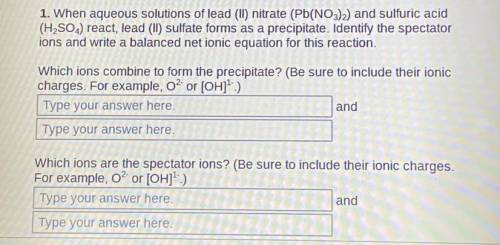
1. When aqueous solutions of lead (II) nitrate (Pb(NO3)2) and sulfuric acid (H2SO4) react, lead (II) sulfate forms as a precipitate. Identify the spectator ions and write a balanced net ionic equation for this reaction.
Which ions combine to form the precipitate? (Be sure to include their ionic charges. For example, O² or [OH]¹-)
2. Which ions are the spectator ions? (Be sure to include their ionic charges. For example, O² or [OH]¹-)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 23.06.2019 14:00
Ahas distinct properties and composition that never vary.
Answers: 1

You know the right answer?
HURRY
1. When aqueous solutions of lead (II) nitrate (Pb(NO3)2) and sulfuric acid (H2SO4) react, le...
Questions



Mathematics, 19.05.2021 17:20

Mathematics, 19.05.2021 17:20



Mathematics, 19.05.2021 17:20

Mathematics, 19.05.2021 17:20

Mathematics, 19.05.2021 17:20


English, 19.05.2021 17:20

Health, 19.05.2021 17:20


Mathematics, 19.05.2021 17:20


Mathematics, 19.05.2021 17:20



Mathematics, 19.05.2021 17:20

Mathematics, 19.05.2021 17:20



