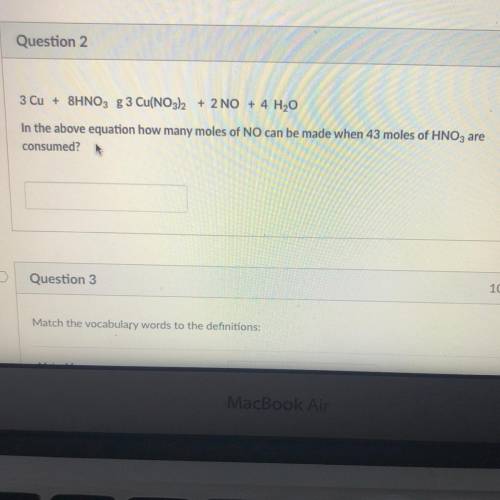
In the above equation how many moles of NO can be made when 43 moles of HNO3 are consumed?
...

Chemistry, 13.04.2021 23:10 arigamez90
In the above equation how many moles of NO can be made when 43 moles of HNO3 are consumed?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1


Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Questions


History, 28.09.2019 22:20


Spanish, 28.09.2019 22:20

History, 28.09.2019 22:20

English, 28.09.2019 22:20


Health, 28.09.2019 22:20




Mathematics, 28.09.2019 22:20





Arts, 28.09.2019 22:20



Mathematics, 28.09.2019 22:20



