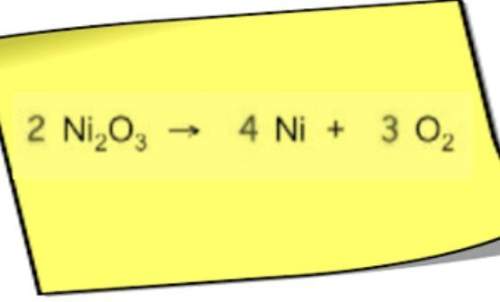
Chemistry, 13.04.2021 01:00 catdog2230
A small iron pan takes 382.0 J to achieve a temperature change of 38.00 ∘C, while a large iron pan requires 2933 J to achieve the same temperature change. Solve to compare the heat capacities of both pans. What is the value of the pan with the higher heat capacity?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
10. translate each of the following chemical equations into a sentence. a. 2 zns(s) + 3 o2(g) -> 2 zno(s) + 2 so2(g) b. cah2(s) + 2 h2o(l) -> ca(oh)2 (aq) + 2 h2(g)
Answers: 2

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2


Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3
You know the right answer?
A small iron pan takes 382.0 J to achieve a temperature change of 38.00 ∘C, while a large iron pan r...
Questions


Medicine, 17.10.2019 02:00


Medicine, 17.10.2019 02:00


Mathematics, 17.10.2019 02:00





Medicine, 17.10.2019 02:00





Medicine, 17.10.2019 02:00


Medicine, 17.10.2019 02:00


Medicine, 17.10.2019 02:00