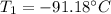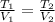Chemistry, 10.04.2021 03:50 lauren21bunch
An ideal gas in a sealed container has an initial volume of 2.80 L. At constant pressure, it is cooled to 18.00 °C, where its final volume is 1.75 L. What was the initial temperature?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2
You know the right answer?
An ideal gas in a sealed container has an initial volume of 2.80 L. At constant pressure, it is cool...
Questions



English, 16.06.2021 22:00


English, 16.06.2021 22:00



Mathematics, 16.06.2021 22:00



English, 16.06.2021 22:00


English, 16.06.2021 22:00


Mathematics, 16.06.2021 22:00




Mathematics, 16.06.2021 22:00