
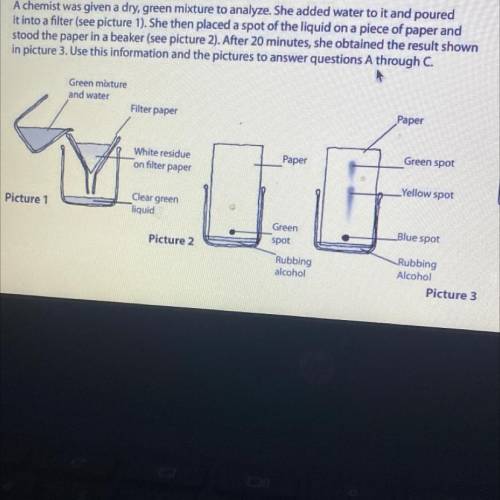
A chemist was given a dry, green mixture to analyze. She added water to it and poured
it into a filter (see picture 1). She then placed a spot of the liquid on a piece of paper and
stood the paper in a beaker (see picture 2). After 20 minutes, she obtained the result show
in picture 3. Use this information and the pictures to answer questions A through C.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
You know the right answer?
A chemist was given a dry, green mixture to analyze. She added water to it and poured
it into a fil...
Questions

History, 18.10.2020 15:01


Mathematics, 18.10.2020 15:01






Engineering, 18.10.2020 15:01

History, 18.10.2020 15:01


Biology, 18.10.2020 15:01

Mathematics, 18.10.2020 15:01

Health, 18.10.2020 15:01

Biology, 18.10.2020 15:01

Spanish, 18.10.2020 15:01

Biology, 18.10.2020 15:01

Social Studies, 18.10.2020 15:01

Biology, 18.10.2020 15:01




