
Chemistry, 08.04.2021 18:40 silviamgarcia
A reaction is first order with respect to reactant A and second order with respect to reactant B. Starting with [A] = 0.175 M and [B] = 0.00250 M, the reaction rate is 2.83 x 10−5 M. S−1. What is the rate constant k? Show your work.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
When curium-242 is bombarded with an alpha particle, two products are formed, one of which is a nudge on. what is the other product
Answers: 3


Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
A reaction is first order with respect to reactant A and second order with respect to reactant B. St...
Questions






English, 26.09.2019 12:30

Mathematics, 26.09.2019 12:30


History, 26.09.2019 12:30

English, 26.09.2019 12:30

Spanish, 26.09.2019 12:30

Mathematics, 26.09.2019 12:30

History, 26.09.2019 12:30




Mathematics, 26.09.2019 12:30



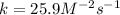
![r=k[A][B]^2](/tpl/images/1246/4928/487f0.png)
![k=\frac{r}{[A][B]^2} \\\\k=\frac{2.83x10^{-5}M/s}{(0.175M)(0.00250M)^2}\\\\k=25.9M^{-2}s^{-1}](/tpl/images/1246/4928/29bac.png)


