
Chemistry, 08.04.2021 08:30 sharkboy3780
A 10.0 mL sample of H2SO4 was exactly neutralized by 23.5 mL of 1.00 M KOH. Calculate the molarity of the acid. H2SO4(aq) + KOH(aq) —> K2SO4(aq) + H2O(l)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1
You know the right answer?
A 10.0 mL sample of H2SO4 was exactly neutralized by 23.5 mL of 1.00 M KOH. Calculate the molarity o...
Questions


Physics, 16.04.2020 16:46

English, 16.04.2020 16:46



History, 16.04.2020 16:46

Biology, 16.04.2020 16:46


Arts, 16.04.2020 16:46

Mathematics, 16.04.2020 16:46

Mathematics, 16.04.2020 16:46

Biology, 16.04.2020 16:46

Mathematics, 16.04.2020 16:47

Biology, 16.04.2020 16:47



Mathematics, 16.04.2020 16:47


History, 16.04.2020 16:47

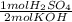 = 11.75 mmol H₂SO₄
= 11.75 mmol H₂SO₄

