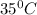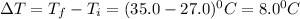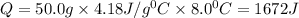Chemistry, 08.04.2021 07:20 david1236544
PLEASE HELP! A piece of metal is heated to a temperature of 50.0°C and then placed in a calorimeter containing 50.0 g of water at 27.0°C. The water temperature increases to 35.0°C. How many joules of heat were transferred from the metal to the water? (Cwater = 4.18J/g•°C)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Apure solvent has a vapor pressure the vapor pressure of a solution. a. equal to b. lower than c. higher than
Answers: 1

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Chemistry, 23.06.2019 10:40
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2
You know the right answer?
PLEASE HELP!
A piece of metal is heated to a temperature of 50.0°C and then placed in a calorimeter...
Questions

Social Studies, 04.08.2019 16:30

Biology, 04.08.2019 16:30

Mathematics, 04.08.2019 16:30

Mathematics, 04.08.2019 16:30

History, 04.08.2019 16:30

Mathematics, 04.08.2019 16:30

Social Studies, 04.08.2019 16:30


Biology, 04.08.2019 16:30

Social Studies, 04.08.2019 16:30



Biology, 04.08.2019 16:30

History, 04.08.2019 16:30

Biology, 04.08.2019 16:30


Chemistry, 04.08.2019 16:30

Chemistry, 04.08.2019 16:30

History, 04.08.2019 16:30

Biology, 04.08.2019 16:30

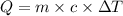
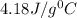
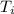 =
= 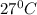
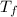 =
=