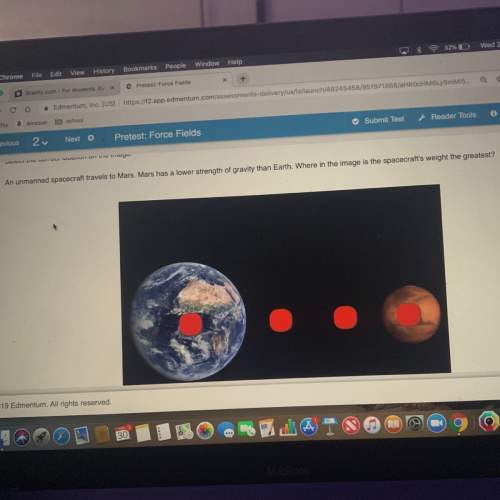
Question 6 of 10
How much energy is required to vaporize 2 kg of gold? Use the table below
an...

Question 6 of 10
How much energy is required to vaporize 2 kg of gold? Use the table below
and this equation: Q = mLvapor
Substance
Latent Heat
Fusion
(melting)
(kJ/kg)
Melting
Point
(°C)
Latent Heat
Vaporization
(boiling) (kJ/kg)
Boiling
Point
(°C)
Aluminum
400
660
1100
2450
Copper
207
1083
4730
2566
Gold
628
1063
1720
2808
Helium
52
-270
21
-269
Lead
24.5
327
871
1751
Mercury
11.4
-39
296
357
Water
335
0
2256
100

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:50
5. how can you decrease the pressure of a gas in a container without changing the volume of the gas?
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
Questions

English, 04.02.2020 16:52

Social Studies, 04.02.2020 16:52

Social Studies, 04.02.2020 16:53

Mathematics, 04.02.2020 16:53

History, 04.02.2020 16:53


Mathematics, 04.02.2020 16:53

Computers and Technology, 04.02.2020 16:53

Biology, 04.02.2020 16:53

Mathematics, 04.02.2020 16:53

Business, 04.02.2020 16:53

Mathematics, 04.02.2020 16:53

Mathematics, 04.02.2020 16:53



Mathematics, 04.02.2020 16:53

Mathematics, 04.02.2020 16:53

Social Studies, 04.02.2020 16:53

Health, 04.02.2020 16:53

Business, 04.02.2020 16:53