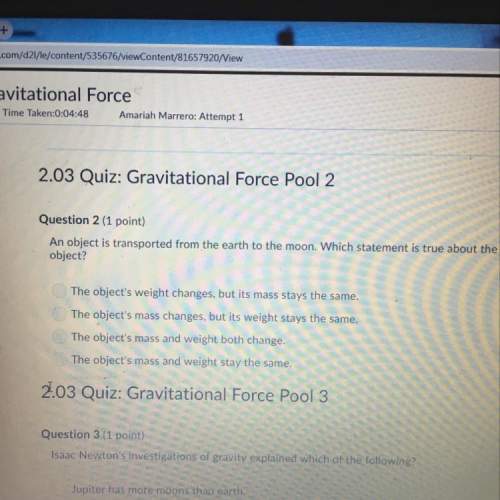
How is the salt formed in a neutralization reaction written in a chemical reaction?
A.
The c...

Chemistry, 01.04.2021 20:40 elyssa34972
How is the salt formed in a neutralization reaction written in a chemical reaction?
A.
The cation is from the base and the anion is from the acid.
B.
The cation is from the acid and the anion is from the base.
C.
The cation and anion both come from the dissociation of water.
D.
The cation and anion may come from either acid or base, depending on their equilibrium constants.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:10
Starch and are common polysaccharide carbohydrates found in plants. sucrose glycogen fructose cellulose
Answers: 3

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Questions



Mathematics, 05.05.2020 18:30

Mathematics, 05.05.2020 18:30




English, 05.05.2020 18:30


Mathematics, 05.05.2020 18:30


Chemistry, 05.05.2020 18:30


Mathematics, 05.05.2020 18:30

Mathematics, 05.05.2020 18:30

Mathematics, 05.05.2020 18:30

Mathematics, 05.05.2020 18:30

Physics, 05.05.2020 18:30