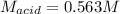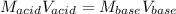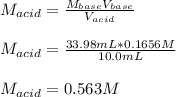Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodium hydroxide solution according to the equation H C 2 H 3 O 2 + N a O H ⟶ H 2 O + N a C 2 H 3 O 2 HCX2HX3OX2+NaOH⟶HX2O+NaCX2HX3OX2 If you require 33.98 mL of 0.1656 M N a O H NaOH solution to titrate 10.0 mL of H C 2 H 3 O 2 HCX2HX3OX2 solution, what is the molar concentration of acetic acid in the vinegar? Type

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3


Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2
You know the right answer?
Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodi...
Questions



English, 31.07.2019 03:00


History, 31.07.2019 03:00



History, 31.07.2019 03:00

History, 31.07.2019 03:00

Biology, 31.07.2019 03:00

Social Studies, 31.07.2019 03:00


Computers and Technology, 31.07.2019 03:00


Social Studies, 31.07.2019 03:00

Business, 31.07.2019 03:00




Mathematics, 31.07.2019 03:00