

Answers: 3


Another question on Chemistry



Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 09:00
Which of the following are in a chemical family a. ca, sc, k b. cu, ag, au c. so, ge, sb
Answers: 1
You know the right answer?
A sealed 1.0L flask is filled with 0.500 mols of I_2 and 0.500 mols of Br_2. When the container achi...
Questions



Chemistry, 18.12.2020 01:00


Biology, 18.12.2020 01:00

Mathematics, 18.12.2020 01:00

Physics, 18.12.2020 01:00




History, 18.12.2020 01:00

Biology, 18.12.2020 01:00




Mathematics, 18.12.2020 01:00


Mathematics, 18.12.2020 01:00


Mathematics, 18.12.2020 01:00

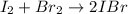
![K=\frac{[IBr]^2}{[I_2][Br_2]} =0.0110](/tpl/images/1228/6485/945be.png)
 (reaction extent) would be:
(reaction extent) would be: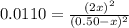
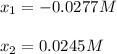
![[IBr]=2x=2*0.0249M=0.049M](/tpl/images/1228/6485/0c444.png)


