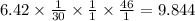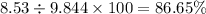Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 6.42 grams of nitrogen monoxide are mixed with excess oxygen gas . The reaction yields 8.53 grams of nitrogen dioxide . nitrogen monoxide ( g ) oxygen ( g ) nitrogen dioxide ( g ) What is the theoretical yield of nitrogen dioxide

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1


Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1
You know the right answer?
Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 6.4...
Questions



Physics, 16.10.2020 16:01



Chemistry, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01



English, 16.10.2020 16:01



Engineering, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01