
Chemistry, 29.03.2021 20:00 mktdonaldson
Which of the following setups will correctly convert 36.45 grams of sodium to moles of sodium?
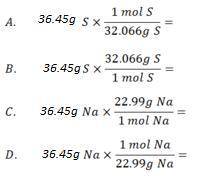

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
Which of the following setups will correctly convert 36.45 grams of sodium to moles of sodium?
Questions





Mathematics, 22.09.2019 22:30

Mathematics, 22.09.2019 22:30

History, 22.09.2019 22:30


History, 22.09.2019 22:30



Biology, 22.09.2019 22:30

Biology, 22.09.2019 22:30



Mathematics, 22.09.2019 22:30

Social Studies, 22.09.2019 22:30

History, 22.09.2019 22:30


English, 22.09.2019 22:30



