

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3
You know the right answer?
For the reaction 2 Al + 3 H2SO4⟶ 3 H2 + H2(SO4)3 how many grams of sulfuric acid, H2SO4, are needed...
Questions

Mathematics, 20.06.2021 14:30

English, 20.06.2021 14:30

Mathematics, 20.06.2021 14:30


Mathematics, 20.06.2021 14:30


English, 20.06.2021 14:30


Chemistry, 20.06.2021 14:40

Chemistry, 20.06.2021 14:40

English, 20.06.2021 14:40


Health, 20.06.2021 14:40

Computers and Technology, 20.06.2021 14:40

Chemistry, 20.06.2021 14:40

Physics, 20.06.2021 14:40




Physics, 20.06.2021 14:40

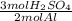 = 2.98 mol H₂SO₄
= 2.98 mol H₂SO₄

