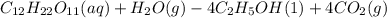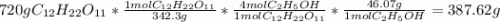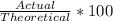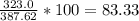6 minutes ago • Chemistry • High School
Ethanol (C2H5OH) is produced from the fermentation of
...

Chemistry, 29.03.2021 06:30 hoolio4495
6 minutes ago • Chemistry • High School
Ethanol (C2H5OH) is produced from the fermentation of
sucrose in the presence of enzymes.
AC
C12H22011(aq) + H2O(g) 4 C2H5OH(1) + 4 CO2(g)
Determine the theoretical yield and the percent yields of ethanol if 720.g
sucrose undergoes fermentation and 323.0 g ethanol is obtained.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Alarge marble is dropped in a graduated cylinder with 35ml of water in it.the water level increases to 49ml.what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3
You know the right answer?
Questions






Social Studies, 02.09.2021 19:40

History, 02.09.2021 19:40

Mathematics, 02.09.2021 19:40


Mathematics, 02.09.2021 19:40

English, 02.09.2021 19:40


Mathematics, 02.09.2021 19:40

Mathematics, 02.09.2021 19:40

Medicine, 02.09.2021 19:40

Arts, 02.09.2021 19:40


Mathematics, 02.09.2021 19:40