
Chemistry, 29.03.2021 02:20 klivingston1012
PLEASE HELP ME YALL I NEED TO GET THIS RIGHT
A chemist wants to extract copper metal from copper chloride solution. The chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (II) chloride. A single replacement reaction takes place. Which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Unbalanced equation: CuCl2 + Al → AlCl3 + Cu (4 points)
a. Approximately 0.36 grams, because copper (II) chloride acts as a limiting reactant
b. Approximately 1.8 grams, because copper (II) chloride acts as a limiting reactant
c. Approximately 0.36 grams, because aluminum acts as a limiting reactant
d. Approximately 1.8 grams, because aluminum acts as a limiting reactant

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 08:30
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
PLEASE HELP ME YALL I NEED TO GET THIS RIGHT
A chemist wants to extract copper metal from copper ch...
Questions

Biology, 25.08.2019 12:30

English, 25.08.2019 12:30


History, 25.08.2019 12:30

Health, 25.08.2019 12:30

Mathematics, 25.08.2019 12:30


Biology, 25.08.2019 12:30


Mathematics, 25.08.2019 12:30




History, 25.08.2019 12:30






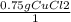 ×
×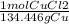 ×
×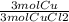 ×
×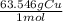 ≈0.35
≈0.35


