
Chemistry, 27.03.2021 19:40 ronniethefun
Sulfuric acid reacts with aluminum hydroxide by double replacement producing aluminum sulfate and water Write chemical reaction. If 30 g of sulfuric acid reacts with 25 g of aluminum hydroxide, identify the limiting reactant?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2

Chemistry, 23.06.2019 08:30
What percentage of energy used in the u.s is produced from fossil fuels
Answers: 2
You know the right answer?
Sulfuric acid reacts with aluminum hydroxide by double replacement producing aluminum sulfate and wa...
Questions




Spanish, 11.06.2021 21:00


Mathematics, 11.06.2021 21:00

History, 11.06.2021 21:00



Biology, 11.06.2021 21:00

History, 11.06.2021 21:00




English, 11.06.2021 21:00


English, 11.06.2021 21:00

English, 11.06.2021 21:00


Mathematics, 11.06.2021 21:00

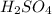 is the limiting reagent.
is the limiting reagent.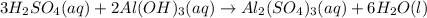

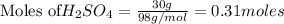
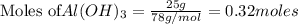
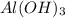
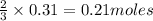 of
of 

