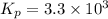Chemistry, 26.03.2021 21:10 dchannakhone84
Ammonia and oxygen react to form nitrogen monoxide and water, like this:
Also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition:
Compound pressure at equilibrium
NH_3 65.3 atm
O_2 7.79 atm
NO 12.1 atm
H_2O 65.8 atm
calculate the value of the equilibrium constant Kp for this reaction. Round your answer to 2 significant figures.
Kp=

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 23.06.2019 03:00
Determine type of reaction & predict the product c3h12+o2 =
Answers: 1

Chemistry, 23.06.2019 05:30
Based on the formulas, select the compounds below that are covalent: kbr sif4 al2o3 co2 naco3 s7o2 pcl3 fe3n2 h2o s2f10
Answers: 3

Chemistry, 23.06.2019 05:50
Aseismic wave is energy released as the result of rock movement along a fault. t or f ?
Answers: 1
You know the right answer?
Ammonia and oxygen react to form nitrogen monoxide and water, like this:
Also, a chemist finds that...
Questions


Social Studies, 27.08.2021 17:40






Mathematics, 27.08.2021 17:40



Biology, 27.08.2021 17:40


Mathematics, 27.08.2021 17:40


English, 27.08.2021 17:40

History, 27.08.2021 17:40

Chemistry, 27.08.2021 17:40

Mathematics, 27.08.2021 17:40

Mathematics, 27.08.2021 17:40

Mathematics, 27.08.2021 17:40


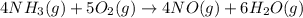
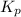 is written as:
is written as:
![K_p=\frac{[p_{NO}]^4\times [p_{H_2O}]^6}{[p_{NH_3}]^4\times [p_{O_2}]^5}](/tpl/images/1224/2313/889d8.png)