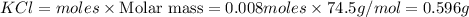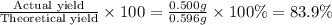Chemistry, 26.03.2021 04:00 jess7kids98
Potassium chlorate decomposes to produce potassium chloride and oxygen gas according to the balanced equation below. If 1.00 g of potassium chlorate decomposes, and 0.500 g of solid potassium chloride is collected, what is the theoretical yield of potassium chloride and the % Yield of potassium chloride

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2

Chemistry, 23.06.2019 03:20
What kind of intermolecular forces act between a hydrogen fluoride molecule and a hydrogen peroxide molecule? note: if there is more than one type of intermolecular force that acts, be sure to list them all, with a comma between the name of each force.
Answers: 1
You know the right answer?
Potassium chlorate decomposes to produce potassium chloride and oxygen gas according to the balanced...
Questions

Mathematics, 29.11.2020 03:40

English, 29.11.2020 03:40


Chemistry, 29.11.2020 03:40



Arts, 29.11.2020 03:40


Biology, 29.11.2020 03:40


History, 29.11.2020 03:40

Social Studies, 29.11.2020 03:40



Spanish, 29.11.2020 03:40

Social Studies, 29.11.2020 03:40



Mathematics, 29.11.2020 03:40

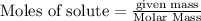
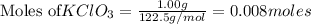
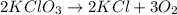
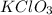 produce= 2 moles of
produce= 2 moles of 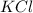
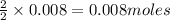 of
of