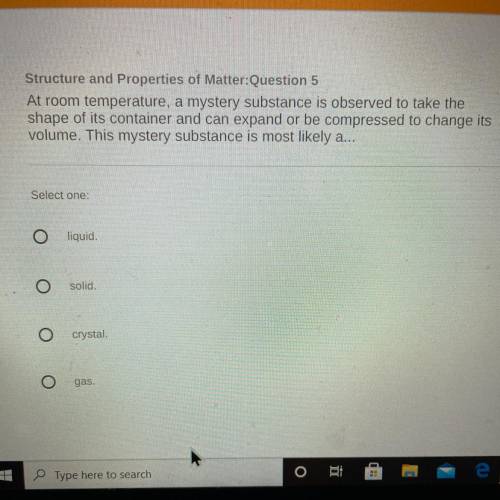
Someone please help. thank you
...

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Alculate the concentration of h3o⁺in a solution that contains 5.5 × 10-5m oh⁻at 25°c. identify the solution as acidic, basic, or neutral.a) 1.8 × 10-10m, basicb) 1.8 × 10-10m, acidicc) 5.5 × 10-10m, neutrald) 9.2 × 10-1m, acidice) 9.2 × 10-1m, basic
Answers: 1

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1


Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
You know the right answer?
Questions


Mathematics, 22.10.2020 23:01


Mathematics, 22.10.2020 23:01

Mathematics, 22.10.2020 23:01

Advanced Placement (AP), 22.10.2020 23:01

Chemistry, 22.10.2020 23:01


Mathematics, 22.10.2020 23:01


Social Studies, 22.10.2020 23:01

Mathematics, 22.10.2020 23:01

Mathematics, 22.10.2020 23:01



English, 22.10.2020 23:01

Geography, 22.10.2020 23:01