What mass of Na is needed to react with
H2O to liberate 566 L H2 gas at STP, accord-
ing to t...

Chemistry, 25.03.2021 03:00 shortyyashaun
What mass of Na is needed to react with
H2O to liberate 566 L H2 gas at STP, accord-
ing to the equation below?
2 Na(s) + 2 H2O(l) → H2(g) +2 NaOH(aq)
Answer in units of g. Your answer must be
within 3.0%

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1
You know the right answer?
Questions

Mathematics, 05.12.2020 14:00

History, 05.12.2020 14:00



Chemistry, 05.12.2020 14:00

SAT, 05.12.2020 14:00

Biology, 05.12.2020 14:00

Advanced Placement (AP), 05.12.2020 14:00

Mathematics, 05.12.2020 14:00

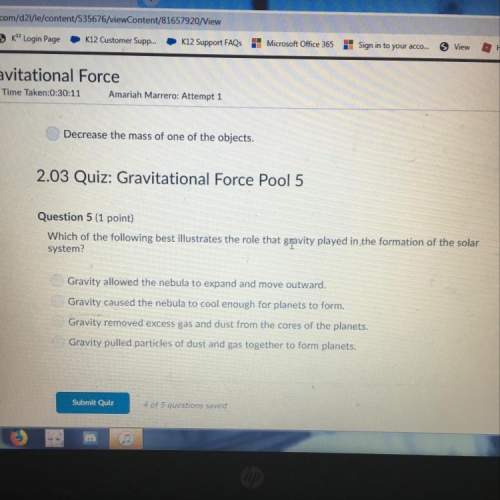
Mathematics, 05.12.2020 14:00




Social Studies, 05.12.2020 14:00

History, 05.12.2020 14:00


Social Studies, 05.12.2020 14:00

World Languages, 05.12.2020 14:00

Mathematics, 05.12.2020 14:00