
Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 00:00
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1
You know the right answer?
What is the oxidation number of cs in cs2o...
Questions


History, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40

Geography, 13.11.2020 06:40

Chemistry, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40


History, 13.11.2020 06:40

History, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40



Mathematics, 13.11.2020 06:40



Mathematics, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40

History, 13.11.2020 06:40

Mathematics, 13.11.2020 06:40

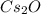 is +1
is +1

