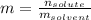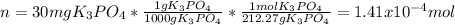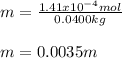Chemistry, 22.03.2021 04:40 trodgers0202
What is the molality of a solution that has 30mg of K3PO4 dissolved in 40mL of water? (The density of water is 1.00 g/mL)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1

Chemistry, 23.06.2019 01:00
Which elements are found in glucose, the product of photosynthesis? a. carbon, hydrogen, and oxygen b. carbon and hydrogen c. carbon, nitrogen, and oxygen d. hydrogen, nitrogen, and carbon
Answers: 2

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

You know the right answer?
What is the molality of a solution that has 30mg of K3PO4 dissolved in 40mL of water? (The density o...
Questions



Mathematics, 08.07.2020 18:01




Mathematics, 08.07.2020 18:01

Computers and Technology, 08.07.2020 18:01






Mathematics, 08.07.2020 18:01