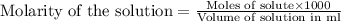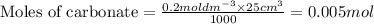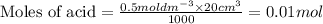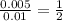A volume of 25cm3 of a carbonate solution of concentration 0.2mol dm-3 was neutralized by 20 cm3 of acid of concentration 0.5 mol dm-3. The mole ratio in which the reactants combine is?
1 mol of carbonate to 1 mol of acid
1 mol of carbonate to 2 mol of acid
2 mol carbonate to 1 mol of acid
3 mol of carbonate to 1 mol of acid

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
You know the right answer?
A volume of 25cm3 of a carbonate solution of concentration 0.2mol dm-3 was neutralized by 20 cm3 of...
Questions

Mathematics, 06.10.2019 04:30




Geography, 06.10.2019 04:30

English, 06.10.2019 04:30


Mathematics, 06.10.2019 04:30

Business, 06.10.2019 04:30

Chemistry, 06.10.2019 04:30




Physics, 06.10.2019 04:30

Mathematics, 06.10.2019 04:30


Chemistry, 06.10.2019 04:30



Computers and Technology, 06.10.2019 04:30