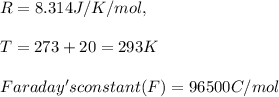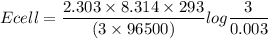A certain metal M forms a soluble nitrate salt M NO Suppose the left half cell of a galvanic cell apparatus is filled with a 3.00 mM solution of M(NO) and the right half cell with a 3.00 M solution of the same substance. Electrodes made of M are dipped into both solutions and a voltmeter is connected between them. The temperature of the apparatus is held constant at 20.0 °C.
1. Which electrode will be positive? What voltage will the voltmeter show?
a. left
b. right
2. Assume its positive lead is connected to the positive electrode. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
A certain metal M forms a soluble nitrate salt M NO Suppose the left half cell of a galvanic cell ap...
Questions

History, 01.09.2021 05:30

Mathematics, 01.09.2021 05:30

Mathematics, 01.09.2021 05:30



Mathematics, 01.09.2021 05:30



Mathematics, 01.09.2021 05:30

English, 01.09.2021 05:30

Mathematics, 01.09.2021 05:30

English, 01.09.2021 05:30

Mathematics, 01.09.2021 05:30

English, 01.09.2021 05:30

English, 01.09.2021 05:30


Mathematics, 01.09.2021 05:30

Spanish, 01.09.2021 05:30


Advanced Placement (AP), 01.09.2021 05:30

![Ecell =\Big( \dfrac{2.303\times R\times T}{nF} \Big)log\Big(\dfrac{[cathode]}{[anode]}\Big)](/tpl/images/1206/7747/f3d33.png)