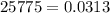Chemistry, 18.03.2021 03:10 hiiamsuperverylong
Methane gas is collected over water at 25 C and an atmospheric pressure of 775 mmHg. What is the dry pressure of the gas at this temperature? (Vapor Pressure of water = 0.0313 atm).

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
A__ is two or more substances that are together in the same place but are not chemically combined
Answers: 3

Chemistry, 22.06.2019 08:00
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1
You know the right answer?
Methane gas is collected over water at 25 C and an atmospheric pressure of 775 mmHg. What is the dry...
Questions




Mathematics, 23.06.2019 17:30

Mathematics, 23.06.2019 17:30


English, 23.06.2019 17:30

Biology, 23.06.2019 17:30

Biology, 23.06.2019 17:30


Mathematics, 23.06.2019 17:30


Mathematics, 23.06.2019 17:30


History, 23.06.2019 17:30