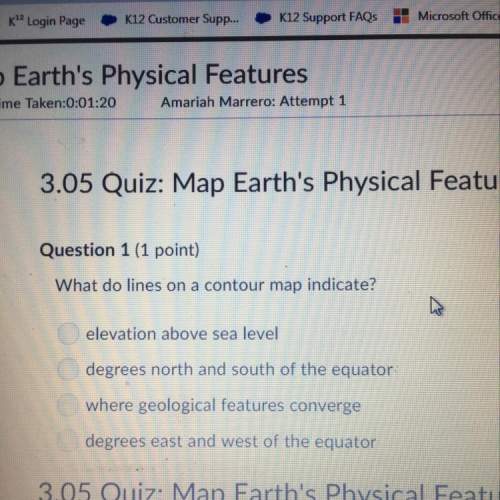
Using the average atomic mass, calculate the mass of 2.949 × 10−5 mol Hg.
Answer in units of g...

Chemistry, 18.03.2021 02:30 jpatte2oye8qv
Using the average atomic mass, calculate the mass of 2.949 × 10−5 mol Hg.
Answer in units of g.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 23:10
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(so4)2·7h2omgso4·7h2o
Answers: 1

You know the right answer?
Questions

Social Studies, 29.07.2020 05:01

Mathematics, 29.07.2020 05:01



Mathematics, 29.07.2020 05:01



Mathematics, 29.07.2020 05:01


Mathematics, 29.07.2020 05:01

Computers and Technology, 29.07.2020 05:01


Mathematics, 29.07.2020 05:01







Computers and Technology, 29.07.2020 05:01