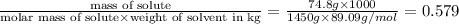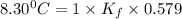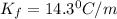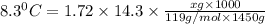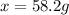Chemistry, 18.03.2021 01:20 Drevei6969
When 74.8g of alanine C3H7NO2 are dissolved in 1450.g of a certain mystery liquid X, the freezing point of the solution is 8.30°C less than the freezing point of pure X. Calculate the mass of potassium bromide that must be dissolved in the same mass of X to produce the same depression in freezing point. The van't Hoff factor =i1.72 for potassium bromide in X.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1
You know the right answer?
When 74.8g of alanine C3H7NO2 are dissolved in 1450.g of a certain mystery liquid X, the freezing po...
Questions

History, 30.10.2019 20:31

Mathematics, 30.10.2019 20:31

History, 30.10.2019 20:31


History, 30.10.2019 20:31



History, 30.10.2019 20:31


Biology, 30.10.2019 20:31



English, 30.10.2019 20:31

Mathematics, 30.10.2019 20:31

Mathematics, 30.10.2019 20:31


History, 30.10.2019 20:31

Biology, 30.10.2019 20:31

History, 30.10.2019 20:31

Mathematics, 30.10.2019 20:31

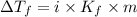
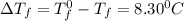 = Depression in freezing point
= Depression in freezing point
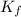 = freezing point constant =
= freezing point constant =