
Chemistry, 13.03.2021 04:50 genyjoannerubiera
Text 8.7 Using the virial equation of state for hydrogen at 298 K given in problem 7 (text 8.6), calculate a. The fugacity of hydrogen at 500 atm and 298 K b. The pressure at which they fugacity is twice the pressure c. The change in Gibbs free energy caused by a compression of 1 mole of hydrogen from 1 to 500 atm. What is the magnitude of the contribution to (c) caused by the non ideality of hydrogen

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
How air particles exert a pressure on the inside of the balloon
Answers: 1

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 23.06.2019 01:00
Which statement best describes isomers? a. isomers are alcohols that have the same functional group. b. isomers have at least one carbon-carbon double bond. c. isomers have the same molecular formula but different structural properties.
Answers: 1
You know the right answer?
Text 8.7 Using the virial equation of state for hydrogen at 298 K given in problem 7 (text 8.6), cal...
Questions

English, 18.12.2020 19:30

Mathematics, 18.12.2020 19:30





Mathematics, 18.12.2020 19:30

Mathematics, 18.12.2020 19:30


Biology, 18.12.2020 19:30



Mathematics, 18.12.2020 19:30

Chemistry, 18.12.2020 19:30


Biology, 18.12.2020 19:30

Health, 18.12.2020 19:30

Biology, 18.12.2020 19:30

Health, 18.12.2020 19:30

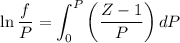
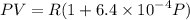 ........(1)
........(1)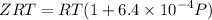
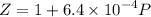
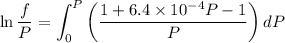
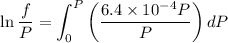
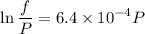
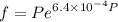
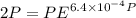
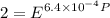
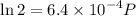
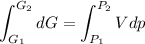
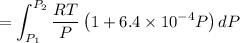
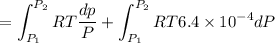
![$\Delta G=R[\ln\frac{P_2}{P_1}+6.4 \times 10^{-4}(P_2-P_1)]$](/tpl/images/1193/5881/be85b.png)
![$\Delta G=8.314\times 298[\ln\frac{500}{1}+6.4 \times 10^{-4}(500-1)]$](/tpl/images/1193/5881/16a41.png)


