Chemistry, 13.03.2021 02:50 bendavidhizar
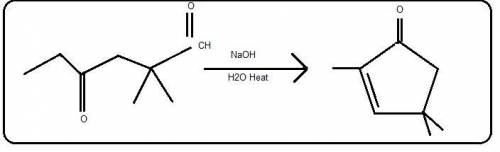
Draw the structure of the product formed when the given compound is heated in aqueous base. The formula for the product is C8H12O. The starting material is a 6 carbon chain where carbon 1 is an aldehyde, carbon 2 has two methyl substituents and carbon 4 is double bonded to an oxygen. THis reacts with N a O H, water and heat.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1
You know the right answer?
Draw the structure of the product formed when the given compound is heated in aqueous base. The form...
Questions



Health, 23.02.2020 05:30


English, 23.02.2020 05:32

Mathematics, 23.02.2020 05:32


Mathematics, 23.02.2020 05:32

Mathematics, 23.02.2020 05:32




English, 23.02.2020 05:33

Mathematics, 23.02.2020 05:33


Mathematics, 23.02.2020 05:33


Mathematics, 23.02.2020 05:34

Mathematics, 23.02.2020 05:34

Mathematics, 23.02.2020 05:34