
Chemistry, 13.03.2021 02:30 eggyhz1980
Please
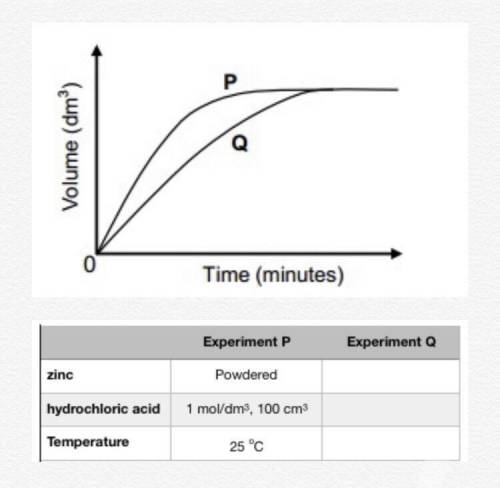
An experiment is carried out to investigate the rate at which hydrogen gas is produced when a given amount of zinc reacts with an excess of a dilute hydrochloric acid solution. Curve P on the graph below shows the volume of hydrogen gas formed when 100 cm3 of a 1 mol/dm3 solution is used. (Reaction rate is indicated by the slope of the graphs.)
Zn + 2 HCl = ZnCl2 + H2
a. Design an experiment that would produce curve Q on the graph below. (Hint: change the experimental parameters so that the rate of gas evolution changes according to the curves). You can also use the table to record your data.
b. What is the implication of the fact that the two curves touch? Explain.
c. Give a reason why the rate of hydrogen production slows down towards the end in both experiments.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which of the following true? a_volcanoes and earthquakes often near the plate boundaries. b_volcanoes occur whereve there are tall mountains. c_earthquakes cause volcanoes in the same location to erupt violently d_volcanoes and earthquakes occur only where plates are colliding with each other
Answers: 2

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2


Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3
You know the right answer?
Please
An experiment is carried out to investigate the rate at which hydrogen gas is produced when...
Questions

Mathematics, 02.07.2019 17:30

Mathematics, 02.07.2019 17:30

Health, 02.07.2019 17:30


History, 02.07.2019 17:30

Social Studies, 02.07.2019 17:30



Geography, 02.07.2019 17:30




Business, 02.07.2019 17:30


Business, 02.07.2019 17:30

Social Studies, 02.07.2019 17:30

Geography, 02.07.2019 17:30





