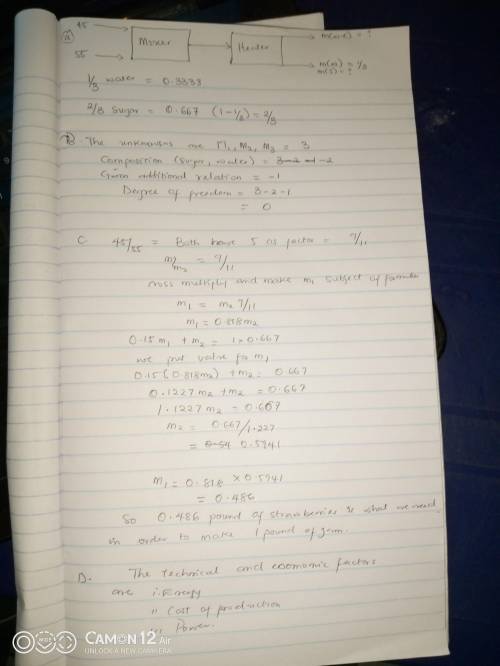Chemistry, 12.03.2021 16:00 Animallover100
Strawberries contain about 15 wt% solids and 85 wt% water. To make strawberry jam, crushed strawberries and sugar are mixed in a 45:55 mass ratio, and the mixture is heated to evaporate water until the residue contains one-third water by mass.
a) Draw and label a flowchart of this process.
b) Do the degree-of-freedom analysis and show that the system has zero degrees of freedom (i. e., the number of unknown process variables equals the number of equations relating them). If you have too many unknowns, think about what you might have forgotten to do.
c) Calculate how many pounds of strawberries are needed to make a pound of jam.
d) Making a pound of jam is something you could accomplish in your own kitchen (or maybe even a dorm room). However, a typical manufacturing line for jam might produce 1500 \mathrm{lb}_{\mathrm{m}} / \mathrm{h}. List technical and economic factors you would have to take into account as you scaled up this process from your kitchen to a commercial operation.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1


Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1

You know the right answer?
Strawberries contain about 15 wt% solids and 85 wt% water. To make strawberry jam, crushed strawberr...
Questions



Mathematics, 18.03.2021 23:30





Mathematics, 18.03.2021 23:30

Mathematics, 18.03.2021 23:30


English, 18.03.2021 23:30





English, 18.03.2021 23:30

Mathematics, 18.03.2021 23:30



English, 18.03.2021 23:30