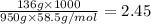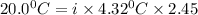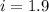Chemistry, 12.03.2021 15:30 Cnolteb5663
When 136g of glycine are dissolved in of a certain mystery liquid , the freezing point of the solution is lower than the freezing point of pure . On the other hand, when of sodium chloride are dissolved in the same mass of , the freezing point of the solution is lower than the freezing point of pure . Calculate the van't Hoff factor for sodium chloride in . Be sure your answer has a unit symbol, if necessary, and round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 23.06.2019 02:00
Which statement is true about the model of the electromagnetic spectrum a: it change the frequencies of light. b: it compare wavelengths of light. c: the color of light waves can be changed using the model. d: the intensities of light waves can be decreased using the model.
Answers: 2

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 2
You know the right answer?
When 136g of glycine are dissolved in of a certain mystery liquid , the freezing point of the soluti...
Questions





Chemistry, 17.11.2021 16:40

English, 17.11.2021 16:40

English, 17.11.2021 16:40



Biology, 17.11.2021 16:40


Mathematics, 17.11.2021 16:40

Spanish, 17.11.2021 16:40


Mathematics, 17.11.2021 16:40

English, 17.11.2021 16:40

English, 17.11.2021 16:40



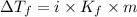
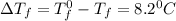 = Depression in freezing point
= Depression in freezing point
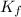 = freezing point constant
= freezing point constant 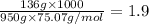
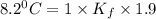
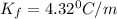
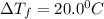 = Depression in freezing point
= Depression in freezing point